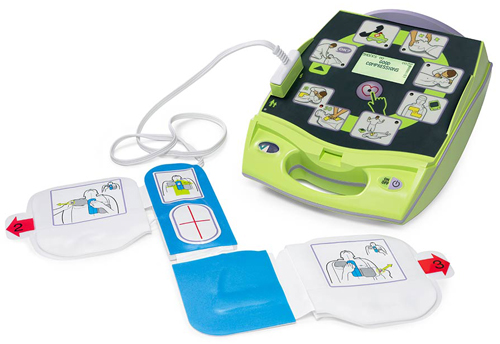
Buying a defibrillator in New Zealand is not an easy task. How do you choose the right one? Will it work when you need it? How can you trust one brand over another? Where is it manufactured?
These are just some of the questions we receive on a daily basis – it’s not just a case of popping down to the local electronics store and buying a new phone!
An AED (defibrillator) is a serious piece of medical equipment – it is a lifesaving device afterall. As such, anyone looking in to buying an AED should do their research thoroughly.
Here are a list of some key questions to ask any company that sells AEDs:
Where is the AED Manufactured?
Some AEDs, especially those made in China, are not subject to the same quality control or safety standards. These “cheap” AEDs also lack the advanced rescue support features of other AEDs, meaning you are actually getting less value for your money and more importantly, an AED that won’t give the patient your best chance of survival.
Is the AED F.D.A. Approved?
FDA approval takes years of research, testing and evaluation and costs millions of dollars to undertake. AEDs that are FDA approved have been thoroughly tested and proven to be safe to use and effective at treating sudden cardiac arrest. Those AEDs which are not FDA approved are likely to provide less support during a rescue, be of lower quality, not last as long and most importantly, not give the patient the best chance of survival.
Does the AED require regular/yearly servicing?
Any AED that requires regular/yearly servicing should immediately ring alarm bells. Not only is this a significant ongoing cost, but it also means you are without your AED while it is sent away to be tested. If a company wants to check on their AED every year, it obviously isn’t up to standard!
Does the AED provide real time feedback to your CPR?
Did you know that a shock is only required approximately 50% of the time? But that 100% of the time you will need to do high quality CPR? As such, you need an AED that provides verbal and visual feedback to your CPR (eg: it tells you to push harder) and one which gives real time live feedback to your rate/speed of CPR – not just a pre-programmed metronome from the factory.